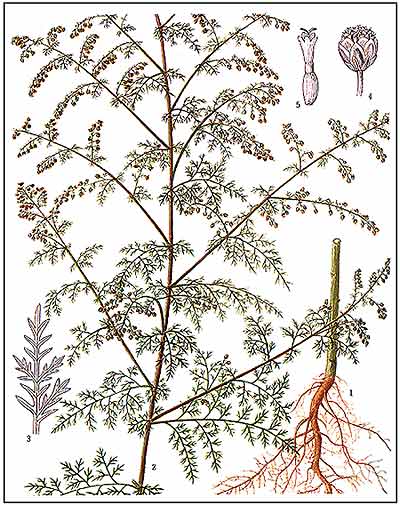
Figure 1: Artemesia annua.
CHING-HAO
and the Artemisias Used in Chinese Medicine
Ching-hao refers to the tops of Artemisia annua (the species name refers to its growth as an annual). The Chinese name (pinyin: qinghao) makes reference to the color green (qing); the dark green leaves at the top of the plant are the most active portion, which might account for this designation. The latter part of the herb's name, hao, indicates this type of plant, one characteristic being a tall stalk; the term is used to depict several other Artemisia species as well. Ching-hao is a common weed in southern China; it is also distributed in temperate regions across the globe, including in Europe and North America. There are numerous medicinal Artemisia species used worldwide, several of them having a reputation for dispelling worms, and hence the group is commonly called "wormwoods." Ching-hao is sometimes referred to by the English common name "sweet wormwood" (also known as sweet annie): the aroma is sweet. It is in the Asteraceae family, and has yellow flowers (see Figure 1).
Ching-hao is markedly bitter in taste, cold in nature, and is classified among the heat clearing herbs, either with those that treat summer heat or those that treat deficiency heat. Summer-heat syndromes are so-named because they usually arise during the summer, when contaminated food and water are prevalent and insect-borne pathogens are transmitted. The syndromes encompassed by this category correspond to various types of infections and parasitic diseases, often accompanied by fever and headache. Deficiency heat is a condition usually accompanied by yin deficiency that involves sweating in response to fever, especially at night. While deficiency-heat syndromes may occur at any time of year, they are more likely during the summer, when the environmental heat contributes to the discomfort.
Ching-hao was described in the Shennong Bencao Jing (1) where it was listed as caohao:
Caohao is bitter and cold. It mainly treats skin bugs, itchy scabs, and malign sores; it kills lice, relieves lodged heat in the joints, and brightens the eyes. Its other name is qinghao.
Based on this brief presentation, ching-hao was primarily used for skin disorders, as a parasiticide, applied topically; a use that is still made of the herb. It was administered internally for rheumatic disorders of the hot type. Three other varieties of Artemisia were also mentioned in the Shennong Bencao Jing: anluzi (Artemisia keiskeanae), baihao (Artemisia argyi; the white hao), and yinchenhao (Artemisia capillaris, yinchen refers to its growth like a carpet of green); the latter is commonly called capillaris. The main parts of each of the descriptions are as follows:
Anluzi is bitter and a little warm. It is non-toxic, treating mainly blood stasis of the five viscera, water qi in the abdomen, abdominal distention, persisting heat, wind-cold-damp impediment, and various kinds of pain in the body.
Baihao is sweet and balanced. It mainly treats evil qi in the five viscera, and wind-cold-damp impediment. It supplements the center, boosts the qi, promotes the growth of hair, is able to turn the hair black, and cures heart suspension [a syndrome involving reduced eating and constant hungering].
Yinchenhao is bitter. It is nontoxic, treating mainly wind-damp-cold, and hot evil qi as well as bound heat jaundice.
For each of these three, but not ching-hao, an immortality benefit is also given, such as "Protracted taking may make the body light, boost the qi, and slow aging." The ability of ching-hao to "brighten the eyes" may parallel these claims rather than being a specific indication for treatment of poor vision: dulling of the eyes is noted as a sign of aging and mortality in some classic texts. The main area of overlap between the four herb descriptions is in the apparent ability of each to treat joint pain. For ching-hao, this indication is heat lodged in the joints; for the other three it is the wind-cold-damp syndrome, which is one that often manifests as arthralgia. Despite these indications, the Artemisias are rarely included in important traditional formulations for treatment of the bi syndromes (the disorders that include arthralgia). Instead, some other applications of the herbs have become more important.
In addition to the Artemisia species of medicinal herbs described in the ancient Shennong Bencao Jing, there are several others used in Chinese medicine. The main ones are the following two (2):
Ching-hao is an ingredient in a small number of well-known traditional formulas. The ones most often referenced in modern texts are the following two indicated for deficiency-heat syndromes, Qinghao Biejia Tang and Qinggu San (3), which are both described below.
Qinghao Biejia Tang (Ching-hao and Turtle Shell Combination)
| Ching-hao | 6 g |
| Turtle shell | 15 g |
| Anemarrhena | 6 g |
| Rehmannia, raw | 12 g |
| Moutan | 9 g |
Turtle shell (biejia) is sometimes referred to in English as tortoise shell; the material used in this formula is differentiated from guiban, which is similarly referenced as either turtle or tortoise shell. Qinghao Biejia Tang, prepared as a decoction (tang), is indicated for advanced febrile disease reaching the blood level and consuming the yin; the formula nourishes yin and clears deficiency heat. Referring to this formula and the role of qinghao, Wu Tong, in his Qing Dynasty text on Warm Diseases, said (11):
One of this formula's excellent features is that it can first enter and later exit. Ching-hao is normally unable to enter the yin aspect, but can be led in by turtle shell; turtle shell, by itself, is unable to exit to the yang aspect [to go to the surface], but can be led out by ching-hao.
This description involves the idea that the pathogen, which started at the surface and then penetrated to the deepest levels of the body, must be engaged and led back out of the body. Turtle shell is heavy and thick and lacks fragrance, so it can easily sink into the deeper levels; ching-hao is light and aromatic, so it can easily float to the surface. The two substances taken together can get into the deep levels, engage the pathogen, and bring it to the surface for release.
Another benefit of ching-hao in this context of clearing deficiency heat is its gentle nature, described by Qin Bowei, one of the founders of modern TCM (6): "Many [heat-clearing] medicinals that are bitter and cold inhibit the stomach and spleen; however, ching-hao is aromatic and pleasing to the spleen, and does not attack the harmony of the middle qi." Ching-hao is one of the six herb ingredients of shen-chu (pinyin: shenqu), a composite of fermented wheat and herbs used to improve the functions of the stomach and spleen and to treat syndromes of summer heat. The related herb capillaris is also deemed very useful in treating heat syndromes (specifically, damp-heat) while benefiting the stomach and spleen. A typical application is treatment of hepatitis with jaundice in persons who have the complication of digestive disturbance with diarrhea.
The other formula in this category of deficiency heat therapies is:
Qinggu San (Powder for Clearing Heat from the Bone)
| Ching-hao | 3 g |
| Turtle shell | 3 g |
| Anemarrhena | 3 g |
| Stellaria | 5 g |
| Picrorrhiza | 3 g |
| Chin-chiu | 3 g |
| Lycium bark | 3 g |
| Licorice | 2 g |
Qinggu San is used for feverish disease in the initial stage when there is pre-existing yin deficiency. It is particularly used when there is high fever, fluctuating fever, and night sweating. The formula is a powder, which is why the dosage description looks so different from that for the previous prescription. There is also a formulation commonly mentioned for gallbladder damp-heat based on use of ching-hao:
Hao Qin Qingdan Tang (Decoction of Ching-hao and Scute to Cleanse the Gallbladder)
| Ching-hao | 6 g |
| Scute | 9 g |
| Bamboo | 9 g |
| Pinellia | 5 g |
| Hoelen, red | 9 g |
| Citrus | 5 g |
| Jasper jade | 9 g |
This formula is aimed at treating damp-heat in the gallbladder and also pervading the shaoyang (liver/gallbladder) channels, particularly when there are alternating fever and chills, with fever more severe than chills. This syndrome may include malaria. In this formula, ching-hao serves a role akin to that of bupleurum in Minor Bupleurum Combination (Xiao Chaihu Tang), a formula with similar therapeutic action used for a related syndrome. Like bupleurum, the aromatic component of ching-hao is said to relieve surface fever and the herb gently regulates the flow of liver qi to improve gallbladder function. Hao Qin Qingdan Tang is widely used in modern China (often modified for specific disorders). It has been administered for treatment of high fever of unknown origin and for cholecystitis (capillaris is more frequently mentioned than ching-hao in traditional formulas for gallbladder diseases).
Two of the Artemisias-aiye and liujinu-are primarily used in treating gynecological disorders, particularly for those cases where there is excessive menstrual bleeding. A well-known formula is Tang-kuei and Gelatin Combination (Xiong Gui Jiao Ai Tang), which includes aiye. Ching-hao is not often selected for this purpose, but is an ingredient in the following traditional prescription for excessive menstrual bleeding in cases of blood heat syndrome:
Qing Jing Tang (Channel Clearing Decoction)
| Ching-hao | 10 g |
| Moutan | 15 g |
| Peony | 15 g |
| Hoelen | 5 g |
| Rehmannia | 15 g |
| Lycium bark | 25 g |
| Phellodendron | 3 g |
The ingredients in this formula, as in the formulas listed above, are directed towards a syndrome of deficiency heat, with rehmannia, lycium bark, and phellodendron being characteristic components of such treatments.
Ching-hao is mainly obtained from Artemisia annua, but this is not the sole source: it is also obtained from A. apiacea, A. scoparia, and other Artemisia species that are collected locally for ching-hao in different parts of China. In Taiwan, Artemisia capillaris (yinchenhao) is used as a substitute, which also illustrates the similarities of these two species that are differentiated in the Materia Medica. Chinese investigations have concluded that only Artemisia annua should be used for ching-hao when its application is dependent on content of artemisinin (see below), which is not found in all species (27).
All of the Artemisias are fragrant herbs, having essential oils comprised of mixed terpenes, with the highest concentration in the flowers. The herb is usually collected when in full bloom. Various species of Artemisias have essential oil contents ranging from 0.15 to 1.0% (30). The primary fragrant component of Artemisia annua is the monoterpene artemisia ketone (see Figure 2), accompanied mainly by other monoterpenes (see Table 1). To retain these compounds in a decoction, it is recommended that the herb be decocted for no longer than 5 minutes (7). These aromatic compounds may contribute some of the antibacterial effects when applied to the skin, and are probably responsible for gallbladder regulating effects of ching-hao (as well as those of capillaris).
Table 1. Essential Oils in Artemisia annua (29). All items specified are monoterpenes, except caryophyllene, a sesquiterpene. Artemisia ketone is unique to Artemisia species, but most of the other components of this essential oil are also found in magnolia bark and flower bud.
| Component | Percent of Essential Oil Fraction |
| artemisia ketone | 66.7 |
| 1,8-cineole (eucalyptol) | 5.5 |
| myrcene | 3.8 |
| linalool | 3.4 |
| caryophyllene | 1.2 |
| pinene | 0.9 |
| camphor | 0.6 |
| borneol | 0.2 |
| others | 17.7 |
One of the largest research efforts undertaken for any Chinese herb was that pursued for the powerful antimalarial effects of ching-hao active components (31). An extensive screening of more than 200 herbs, selected on the basis of their use among 2,000 traditional prescriptions for treating malarial symptoms, was carried out under the direction of Professor Yu Youyou in Beijing. In October of 1971, success was achieved when a new type of sesquiterpene lactone was isolated from Artemisia annua; it was called qinghaosu (su = single ingredient; the term used for isolated active components of herbs). This discovery helped resolve the problem of drug-resistant malarial strains, since those strains were susceptible to treatment by qinghaosu. Considerable work over the next decade was required to bring the laboratory finding to clinical practice. The importance of this finding is illustrated by the fact that six articles appeared in a single quarterly issue of the 1982 Journal of Traditional Chinese Medicine (English) on qinghaosu and its derivatives, describing chemical, pharmacological, and clinical evaluations.
The compound (see Figure 3) was later named artemisinin (alternatively: arteannuin). It is found in the leaves, making up a maximum of about 0.5-0.6% and is highest when the plant is in flower, which is the time traditionally recommended for collection of the herb. However, the amount in different plant samples varies markedly, with some specimens of Artemisia annua having little or no detectable amounts. For example, plants collected in the U.S. typically have very low levels (29). Most of the artemisinin is localized in the upper leaves (see Table 2).
Table 2. Artemisinin levels in different parts of the plant (29).
| Plant Part | Artemisinin as % of total in plant |
| Upper third | 41.7 |
| Middle third | 25.0 |
| Lower third | 22.2 |
| Side shoots | 11.1 |
| Stem, roots, and seeds | 0.0 |
The mechanism of action of artemisinin on the malaria parasite, Plasmodium, has been largely determined. The parasite, which resides in red blood cells, becomes exposed to cytotoxic free radicals and reactive aldehydes produced by the action of artemisinin within the iron-rich environment of the cells. These generated compounds damage the Plasmodium internal membranes and strongly reduce its ability to gain nutritional components required for growth. Thus, the parasites die. The effect is prompt; the parasites are nearly gone within two days when using an effective dosage.
Work has progressed-in the attempt to find more effective antiparasitic compounds-to production of a variety of synthetic derivatives of the sesquiterpene lactones of ching-hao. The main ones, artemether and artesunate, have been subjected to considerable study and development for drug use worldwide.
During the early 1980s, the World Health Organization (WHO) undertook a process of evaluating which antimalarial treatments to recommend for the developing countries where this disease is endemic (28). Artemisinin was considered, but the data available at the time were insufficient to convince authorities of its suitability. There were problems with stability of the drug, questions about an effective method of drug delivery, and concerns about long-term effectiveness (recurrence of disease). The WHO recommendation was to rely on the modern drug therapies, such as chloroquine. The primary user of artemisinin and related compounds for treatment of malaria was Vietnam; the compounds have also been administered in China and Thailand. The malaria problem in Vietnam, which was severe after the Vietnam War, has largely been resolved. This improvement has been attributed, in part, to use of artemisinin as a therapeutic agent (32). It is now proposed that this therapy be used in Africa, where a drug-resistant malarial epidemic is looming: there are about 100 million cases of malaria in Africa each year-90% of all the worldwide malarial infections, with nearly 2 million deaths each year.
Currently, most of the problems with stability, delivery, and efficacy of artemisinin have been resolved. Artemisinin was also declared as effective as chloroquine in monotherapy for malaria. Unfortunately, once the WHO became convinced that artemisinin would be of value, it ran into problems with the Chinese government, which acted, understandably, like a drug company: the negotiations for organizing full scale clinical testing, sound manufacturing practices, and rights for use collapsed (33).
Still, WHO has approved the use of synthetic derivatives, produced in Europe, to be used in cases of drug-resistant malaria, especially the chloroquine resistant malaria that has developed in Brazil and parts of Asia as a result of such widespread use of this drug. The most common method of treating chloroquine resistant malaria is a combination of modern drugs, a method considered more reliable than administering ching-hao compounds, at least in avoiding recurrence of the disease. Thus, even when using artemisinin for drug resistant malaria, it is most often recommended to be given in combination therapy with drugs. For treatment of multi-drug-resistant malaria, artemisinin derivatives (artemether or artesunate) plus mefloquine is recommended; this combination treats the liver stage of malarial parasites, which allows the treatment time to be short (16, 19). A dose of 600-640 mg of the artemether or artesunate plus 1,250 mg of mefloquine over a two day period is often effective.
Artemisinin has been subjected to recent testing in uncomplicated malaria (infection is usually by P. falciparum and the disease is referred to as falciparum malaria). An effort has been made to determine whether this single therapy might be a suitable substitute for the combination drug therapies. Adult dosing at 500 mg/day (first day: 500 mg is given twice for a loading dose) for 5-7 days was tried (15). Parasite clearance usually occurred in the first two days. However, the disease returned with high frequency. In early Chinese clinical studies of artemisinin for malaria, the dosage given was 800-1,600 mg/day for three days, so the dose used in this study was probably too low to assure full clearance of the parasites.
It has been suggested that high dose artemisinin (20 mg/kg body weight; typically more than 1 gram per day) for 2 days be followed up with quinine (30 mg/kg body weight) for 3 days to lower the recurrence rate, which appeared successful in one study (17). In another evaluation, artesunate was administered in cases of uncomplicated malaria using a dosage range from 400-800 mg by oral administration of tablets (14). As little as 400 mg in three days was sufficient to clear parasites and remove symptoms; parasite clearance took place in the first 16 hours. However, 39% of cases treated that way had recurrence within one month; at the highest dosage of 800 mg for 7 days, the recurrence rate was dropped by a factor of 10 to 3.9%. Therefore, high dose treatments, as well as a sufficient course of therapy, may be essential to avoiding recurrence as well as avoiding development of resistant strains (33). By combining artesunate with mefloquine, the recurrence rate can decline to 2% (18), though this level of effect has been claimed for high dose artesunate alone (31). Artesunate is available in injection form and in suppository for rectal administration in patients with advanced disease. One of the proposed uses of it is for treatment of severely debilitated patients in remote areas as an initial therapy before they can be treated by the modern drugs.
Although ching-hao as a crude herb is not as often used today for treating malaria (being replaced by various preparations of the active components), there is information about the method for using it. The dosage of ching-hao recommended to treat malaria is 20-40 grams of the dried herb per day in decoction. In one trial, three days dosing at 24-29 grams per day was successful in curing malaria (recurrence rate not given). Based on a content of 0.5% artemisinin in the herb, these amounts provided about 120-145 mg of the compound, much less than used in the recent trials of the isolated compound or its synthetic derivatives. There may be a group of active compounds in the herb, leading to successful treatment in the dosage range cited; however, recurrence rates may be high.
Presumably, when used in the same amounts that treat malaria, ching-hao can be used prophylactically when traveling in areas with Plasmodium-carrying mosquitoes. If a transfer of the infectious agent occurs via mosquito bite, the daily treatment should eliminate it, without any symptoms appearing. Commonly, the dried, hot water extract is used by travelers, with a dose of 6 grams per day (equivalent, roughly, to 24 grams of herb in decoction). Since treatment time for a case of malaria is just 3-5 days, prophylactic therapy would continue for a few days after leaving an infested area in an attempt to assure clearance of any parasites picked up at the end of the visit. There is no data on the safety of such prophylactic therapy over an extended period of use (e.g., several months). With its pro-oxidant effects, the high doses of ching-hao used in malarial treatments can only be recommended for about 3-6 months, a typical duration for administering ching-hao to treat lupus and other diseases.
Widespread use of ching-hao and its derivatives for prophylaxis (21) or for malaria monotherapy (18) is not recommended; this could simply contribute to evolution of Plasmodium strains resistant to this agent. Use of ching-hao during short-term visits by those who are interested in using Chinese medicine would not be a significant problem in this regard, due to the limited number of people involved. Current recommended prophylaxis for malaria is mefloquine or doxycycline. A problem with any potential malaria prophylaxis is compliance with frequent dosing regimens (23), which may be a drawback for people relying on the herb extracts that are to be administered twice daily in a bulk form (several grams, about a teaspoon full, each time).
In addition to antimalarial effects, artemisinin was found to have promise in treating the parasitic diseases schistosomiasis and clonorchiasis (common in China and Africa, affecting over 200 million people each year) caused by trematodes (blood flukes). Artemether is now being used for prophylaxis against schistosomiasis; in combination therapy with praziquantel it is used to treat the disease (22). Ching-hao is included in effective treatments for leptospirosis, a bacterial disease that usually infects humans from animal waste contaminating water supplies. It has been shown in laboratory studies to inhibit Toxoplasma gondii, an organism that mainly affects persons with compromised immunity, acquired from pets or from eating contaminated meat, and Leishmania major, a protozoa that infects the macrophages. As described earlier, ching-hao was indicated in ancient times for topical treatment of skin parasites. In post-revolutionary China, an antiseptic fumigant was developed using the combination of moxa leaves plus atractylodes (cangzhu). This combination, burned to produce a cleansing smoke, was reported to serve as protection from viruses and bacteria in hospitals; both herbs contributed to the action (4). Recent investigations of the chemical constituents of ching-hao and other Artemisia species have focused on antifungal activity. An oil produced from the herb after its artemisinin was removed, called huanghua oil (yellow flower oil; huanghuahao is another name for ching-hao) was shown to be strongly anti-fungal for all skin fungi tested (26).
Ching-hao appears to regulate T-cell responses and antibody production to inhibit autoimmune reactions (13), with artemisinin being the main active component. Artesunate, which has the same functions as artemisinin, was evaluated in laboratory animal studies and found to suppress allergic contact dermatitis and enhance specific suppressor T-cell activity (24).
Clinically, both ching-hao and artemisinin have been used in the treatment of systemic lupus since 1979, with claimed positive effects in recent trials (12). The dose of artemisinin that has been used clinically for lupus has ranged from 0.2-0.6 grams per day; this corresponds to a dose of ching-hao of about 20-30 grams, the same as used to treat malaria. Treatment time is typically about 3 months. Ching-hao has also been applied in treatment of discoid lupus and was deemed successful (25).
Side effects of ching-hao at normal to high therapeutic doses appear to be rare and mostly involve gastro-intestinal reactions such as nausea, vomiting, and diarrhea (with or without intestinal cramping). In a large study in Thailand comparing high-dose artemisinin derivatives (artemether and artesunate) alone versus in combination with mefloquine, the incidence of adverse effects with the artemisinin compounds was reported to be 34% for loss of appetite, 16% for nausea, 15% for dizziness, and 11% for vomiting; mefloquine greatly increased the incidence of side effects, doubling the rate (20). In a clinical trial comparing artesunate injection with chloroquine and with the combination of quinine and resorcin, no adverse effects of artesunate were reported, while dizziness was a common complaint with the drug therapies (35). Animal studies with high-dose administration of artemisinin had revealed neurological damage, which was initially worrisome, but this appears to be highly species specific and does not affect humans in the dosage range normally used (21). Very high doses of the isolated artemisinin caused liver inflammation in animals and in humans; the human dosage causing this reaction was 10 times the therapeutic dose, given for 3 days (34). Published reports thus far agree that ching-hao, artemisinin, and its derivatives have low toxicity.
March 2002
