Return to ITM Online
EPHEDRINE IN PINELLIA?
The FDA is Charged with Keeping All Ephedrine Out of "Dietary Supplements"
by Subhuti Dharmananda, Ph.D., Director, Institute for Traditional Medicine, Portland, Oregon
BACKGROUND
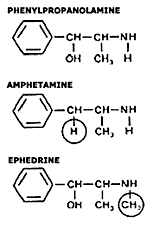
Reports about adverse effects from dietary supplements for weight loss that contain ephedrine have been appearing for more than a decade. These supplement products were developed from research indicating that ephedrine, a naturally-occurring compound mainly found in species of Ephedra, behaves much like the drugs amphetamine and dexamphetamine (Dexedrine), used to stimulate energy, reduce appetite, and promote weight loss. There were studies specifically suggesting that it increased thermogenesis (metabolism) to burn "brown fat," the main fat storage associated with obesity and difficulty in weight loss. The similarity in action between these chemicals is not surprising: ephedrine has only a slight difference from the drugs. Another chemically related compound, phenylpropanolamine (PPA), has been used in sinus remedies until recently: it was withdrawn over safety problems, the same kind of cardiac reactions that apparently occur with ephedrine.
| 
|
In 1997, the FDA attempted to deal with the growing number of adverse reaction reports involving ephedrine in dietary supplements by proposing a limit to the dosage: the proposal was to set a maximum single dose to 8 mg of ephedrine and to limit daily dosing to three times, so that the total daily intake would not exceed 24 mg. Based on their investigations, FDA researchers concluded that this level would be safe for virtually all adult users. Unfortunately, the dietary supplement industry strenuously objected to this restriction: they were typically providing customers with 20 mg (or more) per dose, with three doses per day, in accordance with the research protocols. They understood, correctly, that at 8 mg/dose the product would not be effective for weight loss. To improve the "fat burning" potential of ephedrine and also to provide a greater sensory effect (increased "energy) from taking it, many manufacturers combined ephedrine with caffeine. But, this may have increased the risk for adverse events, and some states banned this combination from sale under their jurisdiction and some even banned the sale of ephedrine supplements entirely.
The FDA came under increasing pressure to regulate ephedrine-containing supplements, which reached its height after Baltimore Orioles pitcher Steve Bechler suddenly died in February 2003. Although there were several factors attributed to his death, the role of ephedrine was prominent in the press reports, and was stated as a definite contributor by the medical examiner. At that time, links of ephedrine to deaths (some of the links being extremely tenuous) were increasing rapidly: there were 88 such reports. Additionally, there were about 1,500 adverse event reports, accounting for about 2/3 of all negative reports about herbs received.
In the meantime, product liability insurers had been excluding coverage for ephedra-containing products, because lawsuits against manufacturers were arising, some of them with multimillion dollar verdicts. The insurers that continued to offer policies increased their prices dramatically, so that only those involved in high-profit weight loss products could afford it.
A few years ago, dietary supplement manufacturers sought to bypass regulations against ma-huang and other Ephedra species, by finding alternative natural sources of the desired compound ephedrine. For example, they turned to an herb mainly used in India, Sida cordifolia, which contains small amounts of ephedrine. This alerted authorities to watch out for substitutions of herb sources where ephedrine might be present. But, the damage was done, and "ephedra-free" or even "ephedrine-free" herbal weight loss products are now widely advertised; few companies want to provide ephedrine-based products at this point.
To bypass regulations and bad publicity on ephedrine, some dietary supplement manufacturers turned to other compounds with similar chemical structures and activities, such as synephrine from bitter orange (Citrus aurantium, Chinese: zhishi). Regulators and insurers have had to play catch-up, finding new herb sources and new substances and making provisions to block their use or to limit the various potential liabilities associated with their use. Recently, the FDA released data it had collected in the past few years that associates 7 deaths and 185 adverse reactions to synephrine-containing weight loss products after receiving a freedom-of-information act request (as opposed to launching its own attack against the products).
People involved with traditional Chinese medicine have been caught in the middle of this competition between dietary supplement producers and those regulating the industry. The commonly used Chinese herb ma-huang is by far the most significant natural source of ephedrine. Its content typically ranges from 0.7 to 1.2% (7,000-12,000 ppm). The dried hot-water extracts-as commonly used by practitioners in their pharmacies as well as by manufacturers of prepared formulas-have 6-8% ephedrine. Although the traditional formulations are not the target of regulations, those who manufactured herb formulas with ma-huang have been forced to either delete the ingredient or take substantial risk in providing the ingredient. Similarly, Chinese herbalists have had to worry about products with bitter orange: zhishi and zhike (chih-shih and chih-ko).
Now, another issue has been raised with regard to yet another commonly used Chinese herb: pinellia (banxia). One of the plants used as a source of this herb was reported to contain ephedrine in a Japanese chemical journal article published in 1978. Even though the content of this component is miniscule-0.002% according to their report-the authors of the article suggested that it was a key active constituent of the herb. In 1996, researchers in Nanjing described their attempt to evaluate the effect of different processing methods for pinellia (ginger juice, alum, or the combination or neither) on the ephedrine content (2). They noted a content of 0.003%, with amounts that seemed to vary somewhat with the processing; using both ginger juice and alum, which is the preferred method, gave the highest amount of ephedrine.

| The situation for Chinese herbalists is complicated by the fact that the report of ephedrine in pinellia has been from tests with the source material Pinellia ternata (referred to as "northern banxia," pictured upper left) while most of the pinellia used in China and in the West is actually from a related plant that has the same uses: Typhonium flagelliforme (referred to as "southern banxia," pictured lower right), which has no ephedrine.
|
Fortunately, as noted above, the ephedrine content reported for Pinellia ternata tubers, raw or processed for the pharmacy, is very low (about 20-30 ppm, or 350 times less than in ma-huang). Pharmacology tests with pinellia (3) have shown it to have sedative, rather than stimulant properties, as might be expected from ephedrine if present in significant quantities. And, testing done for ephedrine thus far by herb companies has mainly shown negative results. For example, from Kaiser Pharmaceuticals (KPC), a letter was sent (4):
This letter is to certify that KPC Products, Inc. has had our Ban Xia single herb granules tested for ephedrine alkaloids. The testing was conducted at the USC School of Pharmacy. The laboratory results show that at a sensitivity of 10 parts per billion, all of the six ephedrine alkaloids are undetectable.
Other companies have gotten similar results (personal communications). Only one company thus far, using products manufactured in China, has claimed any positive tests for ephedrine in pinellia samples.
Unfortunately, the public demands on the FDA are for ephedrine-free products. The FDA has stated that its regulations on dietary supplements are not intended to influence herb prescribing by health professionals or to affect Asian herbal products. Nonetheless, at least one Chinese herb product manufacturer, rather than awaiting adverse rulings or publicity, has taken pinellia out of their products. In the end, testing for ephedrine and/or reliance on southern banxia for pinellia will resolve most of the problem, though the common name "pinellia" which has been applied all along, may still cause some people to worry about its potential content of ephedrine.
ADDENDUM: SAFETY OR SECURITY?
Simply by having the media raise the issue of ephedrine in Chinese herbs (ma-huang and pinellia), providers are put in a somewhat difficult situation. The biggest potential problem is explained by the following theoretical situation.
A practitioner prescribes a Chinese herb formula to a patient. A few days later, the patient has a serious cardiac event, perhaps a heart attack, or another circulatory even, such as a stroke. The patient's wife talks with the doctor about this traumatic event and says he seemed entirely healthy up to this point, only some minor problems like sinus allergies, and too young to be suffering from such a serious disorder. The doctor might say that his last check-up didn't reveal anything unusual. Has he been doing anything different? Well, he just went to an acupuncturist for his allergies and was prescribed some Chinese herbs. Chinese herbs! Well, there have been a lot of negative reports on those, they can be quite dangerous. Better check the label of what he got.
She goes home, reads the label, which isn't very informative, but goes on the internet to read up on the ingredients. According to the internet site, one of the ingredients (could be pinellia, ma-huang, or bitter orange) is said to have the potential to cause severe cardiac reactions, even death. She calls the doctor to report that, in fact, his herb supplement had one of these ingredients. The doctor says: that could have been a contributing factor.
The practitioner receives an agitated phone call about this, and, soon after, the practitioner and the product manufacturer are soon served with a lawsuit alleging that they provided herbs that are known to be potentially dangerous, and did not provide adequate warnings that severe cardiac reactions and even death could result from taking the supplement. The suit alleges that the parties involved should have known that these substances were dangerous and failed to react responsibly. In this scenario, the fact that the herb involved might contain a small amount of the substance of concern is not a deterrent to the legal actions, because there is no study proving that, below a certain level, the substance is safe.
If this imaginary scene seems unlikely, consider the news report in the Oregonian about a person living in the town of Aloha (this story was read after the scenario above was written). The headline (front page of the local news section, January 12, 2004) reads: "Aloha stroke victim sues maker of ephedra product." The story describes a 35-year-old man who had been taking a popular ephedrine-containing weight loss product for "about a month" (it appears from the story that it was just short of a month). The product gave him more energy and helped him lose some excess weight, he said. He said also that doctors couldn't figure out what caused the stroke. "About a month later, his neurologist raised concerns about the ephedra-based supplement. His wife also started noticing TV news reports about the dangers of ephedra." He and his wife are suing the manufacturer, product distributor, and the gym where he bought the product, with a claim for $2.6 million dollars in damages. The lawsuit was filed the same day that the U.S. FDA announced its intention to ban ephedrine in dietary supplements.
Did the ephedra in this product contribute to the stroke? Perhaps, though it would seem strange to many involved with Chinese herbs who have prescribed, and themselves used, ma-huang containing formulas for years. Despite the seemingly large number of adverse event reports for the dietary supplements, it is a small number compared to the millions who have used the products over the years. Those who have looked into the adverse event reports carefully find many of them having insufficient evidence for a link to the ephedrine in the products, suggesting that the incidence rate is not as great as it seems (which doesn't rule out rare serious adverse effects). In this case, the story noted (without suggesting any possible link), that the individual who suffered the stroke "rode his bike 25 miles to and from work nearly every day" and weighed 240 pounds. Perhaps the exercise, plus a congenitally weak vessel in the brain, caused the reaction. But, this would be the subject for experts to debate as a legal case drags on for years.
A significant problem is evident: there is increasing suspicion that "natural" supplements are potentially dangerous, with repeated reference to Chinese herbs (including issues such as drugs and heavy metals in imported patents, and safety concerns about the herbs mentioned above and others, such as the ones containing aristolochic acid). News stories about adverse reactions occur frequently; the widely quoted magazine, Consumer Reports, produced an alarming article on the subject in its May 2004 issue, naming bitter orange and aristolochia, among other herbs.
It is not a workable solution to remove ingredients from formulas every time a concern is raised. As mentioned, some product manufacturers have already removed pinellia from their products, perhaps without even confirming the botanical origins or the ephedrine content. The reason this is not a workable solution is that traditional Chinese herbal medicine becomes an untenable practice if several key herbs are removed. Already, formulations must do without stephania, akebia, aristolochia fruit, asarum, and ma-huang, while many patients and practitioners worry about negative stories concerning ginseng, licorice, and other central ingredients. Magnolia bark has already been a target of insurance company concerns. If one were to also eliminate pinellia, chih-shih, chih-ko, and similar ingredients, few traditional formulas could be produced.
On the other hand, it is not prudent to simply take a wait-and-see attitude: this happened with the problem of aristolochic acid, where ability to test for the ingredient was virtually absent, but removal of the herbs did not seem to be the correct action based on traditional practice and absence of reports of adverse reactions to the herbs from China, where they were used extensively. In fact, continued availability of herbs with aristolochic acid had serious consequences.
Therefore, it is necessary to take each warning seriously, but to take steps that are consistent with the combined circumstances under which the issue has been raised. In the case of pinellia, the ephedrine content of Pinellia ternata is so low, and the source material is so often not Pinellia ternata but Typhonium, that there is room to deal with this issue effectively without removing the herb from Chinese formulas.
In addition to having tests conducted for ephedrine content of pinellia, practitioners should provide information to patients that is sufficiently detailed to allow the patient to participate in a decision about whether or not to use the product. This information could include the fact that pinellia used by Chinese herbalists in the U.S. is usually from the related plant source Typhonium, and is free of ephedrine, and that where Pinellia ternata is used, there is only a trace of ephedrine, and (in most cases), pinellia only makes up a small fraction of the total formulation, but the product may not, in this case, be completely ephedrine-free.
Note: For additional information on these subjects, see the ITM herb safety reports, including these:
Safety Issues Affecting Herb: The Case of Ma-huang
Synephrine: Is Chih-shih Dangerous?
Safety Issues Affecting Herbs: Herbs that may Increase Blood Pressure
Other reports describe concerns raised about magnolia bark, ginseng, Aristolochia plants, kava, usnea, dictamnus, and asarum. A report about the nature, constituents, and botanical relationships between pinellia and typhonium is titled: Pinellia, Arisaema, Acorus, and Typhonium.
REFERENCES
- Oshio H, Tsukui M, and Matsuoka T, Isolation of l-ephedrine from pinellia tuber, Chemical and Pharmaceutical Bulletin 1978; 26: 2096-2097.
- Wu H, et al., Effect of ginger-processing on l-ephedrine contents in Rhizoma Pinelliae, Journal of Chinese Herbal Drugs 1996; 21(3): 157-158, 190.
- Mao SJ, Wu LY, and Cheng LP, Sedative and anticonvulsive effects of differently processed Rhizoma Pinelliae, Journal of Chinese Herbal Drugs 1994; 19(4): 218-220, 256.
- Schuler M, Letter dated April 30, 2004.
May 2004

