|
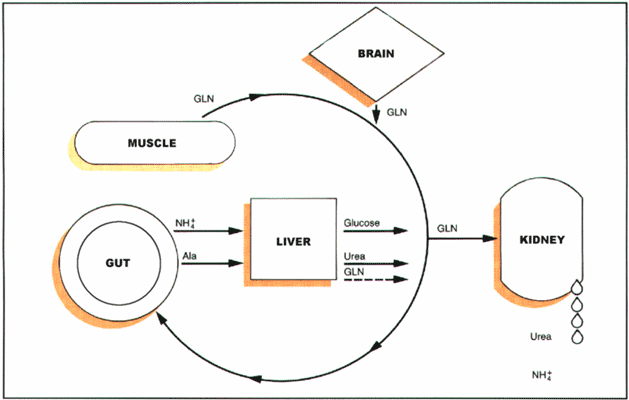
AMINO ACID SUPPLEMENTS I: GLUTAMINE with Reference to the Related Compound Glutamate BACKGROUNDGlutamine, and the closely-related compound glutamate, are two amino acids that are critical to human health. In humans, animals, and plants, glutamine and glutamate are transformed into each other as part of numerous physiological processes. The roles of these amino acids in human diseases were intensively researched during the past decade and continue to be the subject of scrutiny. One area of interest is their potential health effects when they are provided in addition to the normal dietary intake that is already several grams per day. Glutamine has been recommended by nutritionists as a dietary supplement for several serious disease conditions and there is growing research support for this action. Hospital dietitians are aware of the value of administering glutamine in parenteral nutrition (IV nutrition) for critically ill patients, and especially for patients who have had intestinal surgery, but physicians rarely recommend glutamine supplementation in other situations for which it may be indicated. Therefore, it is valuable for other health care providers to be familiar with the over-the-counter availability and uses of glutamine for supplementation purposes. Glutamate is also included in parenteral nutrition and is the major amino acid in certain protein-rich foods, such as eggs. Glutamate is a widely used flavoring agent, best known in the form of its sodium salt, monosodium glutamate (MSG). Harmful effects of localized high concentrations of glutamate in certain neurological disease conditions, such as multiple sclerosis and ALS, have been mentioned in recent literature. This has raised concerns about the safety of administering glutamine (which can be converted to glutamate), consuming foods with added glutamate, or including glutamate in the parenteral nutrition for such patients. In addition, there has been much concern expressed about possible allergic, or other reactions, to glutamate used as a flavoring agent. Therefore, it is valuable to understand the nature of its actions, its relationship to glutamine used as a supplement, and the current status of the relevant research. GLUTAMINEGlutamine is the most abundant of the amino acids in the human body. Its main storage site is in the musculature, where about 60% of all the unbound amino acids are glutamine (glutamine makes up a smaller percentage of muscle protein, the main bound form). Glutamine has been called a "conditionally essential" nutrient (1-6), because it is non-essential in normal situations (manufactured by the body in adequate quantities; not required in the diet), but in severe illness or injury becomes insufficient (there is then a need for supplementation from the diet or other sources). Dietary glutamine is especially prevalent in wheat and beans and in protein isolates, such as those used for making nutrition bars and beverages: glutamine makes up 6-9% (by weight) of soy protein and milk protein (casein, whey) isolates. Glutamine is manufactured in the body from glutamate and ammonia by the enzyme glutamine synthetase; the process takes place mainly in the skeletal muscles. The connection of glutamine to the musculature is of special interest. The amount of glutamine in reserve for release as needed is directly related to muscle mass: more muscle mass means more glutamine is available for metabolic processes. It is possible that one of the benefits of muscle-building exercise for good health is the increased availability of glutamine during times of stress. Under conditions of metabolic stress, including injuries, illness, and even severe emotional distress, the level of glutamine in the body declines markedly, which is thought to adversely influence resistance to infectious diseases. Persons who maintain a relatively large muscle mass may have a greater ability to withstand and recover from stressful events. Chronic illness and lack of exercise work together in a vicious cycle: poor health makes it more difficult to exercise, leading to lower muscle mass and lower glutamine stores, contributing to a higher incidence of health problems and slower recovery. Aside from lack of exercise (which may be a lifestyle choice or the result of paralytic or debilitating diseases and injuries), there are several muscle wasting diseases, including cancer, AIDS, and pulmonary obstructive diseases. In such cases, glutamine levels can be insufficient and contribute to the overall pathology. This deficiency of glutamine, related to muscle wasting, may be partially rectified by consuming extra glutamine daily to replace the muscular manufacture of this amino acid. Additionally, glutamine administration at high doses may be associated with increased production of growth hormone, which contributes to increasing muscle mass. The reasons that the body retains such high levels of glutamine are not fully known, though the number of biochemical reactions that take place in the body involving glutamine is quite large. Amino acids are defined, in part, by the presence of a nitrogen group in a small acidic molecule. Glutamine has two nitrogen groups, one of which, a terminal NH2, is easily separated and transferred to other molecules (leaving glutamate as the amino acid, which has an oxygen atom in place of the extra nitrogen group). Glutamine has been described as the most important circulating nitrogen shuttle, accounting for about one-third of all amino acid nitrogen transported by the blood. By contributing to the formation of many useful compounds, the circulating glutamine brings metabolic fuel to the various organs (see Figure 1). It also transports ammonia in a non-toxic form for excretion (the ammonia is linked to glutamate to form the glutamine). In the kidneys, glutamate is the end product when the ammonia is released (to yield urea) under the control of the enzyme glutaminase. One of the well-established roles of glutamine in human health is its contribution to the integrity of the intestinal mucosa. This role is partly related to the fact that glutamine is a critical nitrogen source for rapidly dividing cells, such as those that line the gastro-intestinal tract. The principal location of glutamine consumption in the body (i.e., where it is broken down to glutamate at the highest rate) is in the small intestine. During times of stress, the small intestine responds by utilizing more glutamine and by more efficiently transporting glutamine that has been ingested. One of the damaging effects of cancer chemotherapy is the inhibition of these cells that line the gastro-intestinal tract, leading to a variety of adverse symptoms, such as nausea, loss of appetite, and reduced absorption of nutrients. Glutamine has been used therapeutically to protect against the toxic effects of methotrexate and other chemotherapy drugs. Glutamine is a useful adjunct to patients undergoing bone-marrow transplant procedures (e.g., intensive chemotherapy to prevent rejection), for which it is reported to improve recovery, reduce infections, and minimize complications. Intestinal surgery, such as that done to remove tumors or to remove ulcerated portions of the intestines, greatly damages the normal mucosal production, leading to slow healing. Glutamine has been used as a means of aiding recovery from intestinal surgery. Small intestine disorders, such as ulcers and bleeding (as occurs with Crohn's disease), may also benefit from extra glutamine administration. Glutamine should not be considered a cure-all for gastro-intestinal disorders, or any of the other conditions for which it has been indicated. Rather, if there are indications of glutamine deficiency (e.g., low muscle mass, high levels of stress, persisting and/or severe intestinal disorder), then it is a reasonable therapeutic strategy to apply. Glutamine is also considered important for the maintenance of the renal tubules, contributing to the healthy function of the kidneys. Glutamine's metabolic activities in the kidneys helps assure elimination of acids from the blood. Although not yet subject to research, kidney-damaging drug therapies might be made more safe by providing extra glutamine, especially in patients who have low muscle mass. As a fuel for rapidly dividing cells, glutamine makes a contribution to the immune system, especially in the rapid production of white blood cells during an infection. The immune system impairment that occurs after severe burns and surgical interventions is thought to be partly due to a rapid decline in glutamine that is part of the stress reaction, resulting in lowered immunological responsiveness. Supplementation of glutamine has been proposed as a means of preventing this consequence of injuries. Glutamine is already a therapy for patients with multiple organ failure and for multi-trauma patients. It has been reported that patients who require IV-feedings due to advanced disease conditions show improvement in mood when glutamine is included in the solution. This change may be due to the improvements in overall physiologic conditions brought about by the higher glutamine levels. The powerful antioxidant glutathione is comprised of three amino acids: glutamate, cysteine, and glycine. Glutamine is described a "glutathione-sparing" agent, helping to maintain adequate levels of glutathione by providing adequate glutamate for its production. Glutathione deficiency tends to arise with glutamine decline (e.g., with muscle wasting) and is compensated for by administering glutamine. Glutathione is thought to contribute numerous protective effects from the adverse effects of oxidative stress and has been proposed, when administered as a supplement, to help inhibit the development of cancer, gastric ulcers, and other diseases. While glutathione taken as a supplement is very expensive and poorly absorbed, glutamine is far less expensive and easily absorbed. Studies have shown that glutamine administration leads to higher glutathione in patients undergoing surgery and reduced incidence of infections (21). Dosage, Absorption, and MetabolismThe usual dose of orally administered glutamine for the various applications mentioned above is 0.5-0.57 grams/kilogram of body weight, which is about 25-30 grams per day for an adult who has low muscle mass (e.g., body weight of only 50 kg, about 110 pounds). Recommended adult doses of glutamine taken orally range from as little as 5 grams per day (roughly matching the dietary levels) to about 40 grams per day (higher doses become impractical to administer and may provide no further benefits). The dosing is partly determined by body weight, with doses of 0.1-0.8 grams/kg being given according to various recommendations; the largest amounts are usually reserved for cases where there is little dietary glutamine and high need for it, such as after intestinal surgery when the patient cannot eat ordinary foods. Because glutamine is efficiently absorbed in the small intestine, blood levels reach a peak within an hour after ingestion. IV administration of glutamine is only utilized when parenteral feeding is already required. Glutamine is available as a bulk powder that is essentially tasteless. Quantities of 5-15 grams at a time can be consumed easily by mixing the powder in water, juice, or a blender drink. Encapsulated glutamine is probably ineffective due to the low dosage obtained by that method of administration (encapsulation also makes the material more expensive to use). Glutamine is metabolized to other amino acids, including glutamate, alanine (the second most abundant amino acid in skeletal muscle), citrulline, and arginine; in leukocytes, it can ultimately be metabolized to carbon dioxide. Following administration of glutamine, the increased blood content of the various amino acids that arise from glutamine metabolism return to baseline after a few hours (about four hours with high dose administration). It is possible that some liver or kidney diseases may lead to difficulties in metabolism of glutamine, so that administration of the substance in high doses should only be undertaken after adequate evaluation of the patient's condition and with careful monitoring of the responses to the glutamine administration. When administered at high doses over a long period of time (e.g., for several consecutive days), the body's own production of glutamine declines in compensation. As a result, the blood levels of glutamine, though higher than they were prior to supplementation, do not rise beyond a certain point because of the compensation by lower production rates. Glutamine and CancerThe role of glutamine in cancer has been a topic of recent interest. On the one hand, glutamine seems to be the ideal treatment for the cachexia that accompanies tumor growth and the adverse reactions to chemotherapy. On the other hand, it has been found that tumor cells are capable of efficiently transporting glutamine and that this is one of their major respiratory fuels. Therefore, the question has been raised as to whether administering glutamine might not also be beneficial to the cancer. Thus far, all indications are that glutamine is useful as part of cancer therapy (7, 8). In animal models, glutamine administration does not enhance the cancer growth. In order to help assure this outcome in human patients, chemotherapy can be given at the same time. As the cancer develops, it can compete with the rest of the body for the glutamine supply. The body's metabolism is forced to shift into high absorption of dietary glutamine and production of glutamine by the liver and other tissues. Attempting to block tumor growth by starving it for glutamine is not practical, as the body will be starved of glutamine first. Using glutamine analogs that will be taken up by the tumors in place of glutamine but which fail to nourish the tumor have been developed, but have not been found to be viable treatments as yet. Instead, it has been suggested that cancer patients be given supplemental glutamine so that their body levels can be maintained. Summary of Applications of GlutamineThe potential applications for orally administered glutamine include:
GLUTAMATEGlutamate is an amino acid similar to glutamine, but having a charged oxygen atom (making the compound negatively charged) in place of one of the neutral nitrogen groups. It is also described as glutamic acid, with glutamate being the free form (usually associated with one or more metals, such as sodium or potassium, to compensate the negative charge, making it a "salt") and glutamic acid being the term for the bound form, in complex proteins. As described above, glutamine and glutamate are transformed back and forth during normal body metabolism. In the blood, glutamate is present at a level about 25% that of glutamine. It is the third most abundant amino acid in the blood; taurine is second (taurine is found in the free form, but not in proteins). Unlike glutamine, there is little storage of glutamate in the muscles. In natural foods, glutamic acid is much more common than free glutamate, so the analysis of dietary amino acids is described in terms of glutamic acid content. It is found in all protein-containing foods, especially in animal products, such as cheese (cottage cheese provides about 6 grams/cup) and meat (beef round steak provides 13 grams/pound), but also in protein-rich nuts (almonds provide 8 grams per cup) and beans. Monosodium glutamateThe glutamic acid containing foods are broken down to release glutamate in the digestive tract. Glutamate can be present in relatively large quantities in certain meals in the form of a flavoring agent, monosodium glutamate (MSG), which has been in widespread use for nearly a century. Glutamate is also a significant component of hydrolyzed vegetable proteins (proteins broken down to release some amino acids) used as flavorings. MSG enters some diets far more than others; for example, in the diets of southeast Asia (e.g., Thailand, Hong Kong), the daily intake level for this compound reaches about 5 grams, contributing about 4 grams of glutamate to the other dietary sources. The presence of sodium in MSG is not relevant to the material's contribution of the amino acid glutamate. It does increase the total sodium in the diet and it is relevant to the flavoring action of the compound. Because glutamate is a normal component of foods and a major amino acid in the body, there is no reason to expect any toxicity from consuming glutamate as a flavoring in the form of monosodium glutamate. However, reports of possible adverse responses to MSG, which led to the description of "Chinese restaurant syndrome" (a variety of discomforts described by some non-Oriental people after eating at a Chinese restaurant), gave rise to considerable research into its effects. Although a few early studies seemed to confirm the existence of a reaction (later dubbed the MSG symptom complex, since the compound can be found outside of Chinese restaurants and the reaction in the restaurants might be due to something else), almost all of the well-designed modern studies have found little or no effect. In a frequently cited evaluation of acute toxicity, huge single doses of 10 grams of MSG administered to volunteers produced no evidence of toxicity. As a result of this and long-term studies involving relatively high doses that appeared safe, there have been no limits placed on the quantities that can be used in food preparations (which are usually in amounts of only a few hundred milligrams per serving). Rare hypersensitivity to MSG has not been ruled out, but repeated double-blind studies have shown little or no reaction to MSG among persons who considered themselves MSG sensitive based on their own interpretations of experiences with foods. In particular, it had been suggested that MSG could worsen asthma and that it caused various allergy reactions, including skin rashes. A collection of recent research reports from around the world was published in the April 2000 issue of the Journal of Nutrition, and several other reports appeared in the Journal of Allergy and Clinical Immunology during the period 1998-2000. Among the conclusions were these: ...MSG has been suggested to cause postprandial symptoms after the ingestion of Chinese or Oriental meals. Therefore, we examined whether such symptoms could be elicited in Indonesians ingesting levels of MSG typically found in Indonesian cuisine....The study used a rigorous, randomized, double-blind, cross-over design. The occurrence of symptoms after MSG ingestion did not differ from that after consumption of the placebo (Yogyakarta, Indonesia; 13). ...MSG challenges in subjects with and without a perceived sensitivity to MSG failed to induce signs or symptoms of asthma. Therefore, in view of the poorly conducted studies that proposed that MSG induced asthma and the subsequent studies that failed to confirm those findings, it is important to maintain a healthy skepticism about the existence of MSG sensitivity in individuals with asthma (La Jolla, California; 15). ...The existence of MSG-induced asthma, even in history-positive patients, has not been established conclusively (La Jolla, California; 16). ...MSG-induced asthma was not demonstrated in this study. This study highlighted the importance of adequate baseline and control data and indicated that such a rigorous protocol for individual assessment is feasible (Melbourne, Australia; 17). ...In patients with chronic urticaria, the incidence of reactions to any additives, including MSG, is unknown....The dose of MSG given was 2,500 mg....We conclude, with 95% confidence, that MSG is an unusual (less than 3% at most) exacerbant of chronic idiopathic urticaria (La Jolla, California; 14). ...Because human studies failed to confirm an involvement of MSG in "Chinese Restaurant Syndrome" or other idiosyncratic intolerance, the Joint FAO/WHO Expert Committee on Food Additives (JECFA) allocated an "acceptable daily intake (ADI) not specified" to glutamic acid and its salts. No additional risk to infants was indicated. The Scientific Committee for Food (SCF) of the European Commission reached a similar evaluation in 1991. The conclusions of a subsequent review by the Federation of American Societies for Experimental Biology (FASEB) and the Federal Food and Drug Administration (FDA) did not discount the existence of a sensitive subpopulation, but otherwise concurred with the safety evaluation of JECFA and the SCF (Rome, Italy; 10). ...Results of surveys and of clinical challenges with MSG in the general population reveal no evidence of untoward effects. We recently conducted a multicenter, double-blind, placebo-controlled challenge study in 130 subjects (the largest to date) to analyze the response of subjects who report symptoms from ingesting MSG. The results suggest that large doses of MSG given without food may elicit more symptoms than a placebo in individuals who believe they react adversely to MSG. However, the frequency of the responses was low and the responses reported were inconsistent and not reproducible. The responses were not observed when MSG was given with food (Boston, Massachusetts; 9). ...Controlled double-blind studies have failed to establish a relationship between Chinese Restaurant Syndrome and ingestion of MSG, even in individuals reportedly sensitive to Chinese meals, and MSG did not provoke bronchoconstriction in asthmatics. Thus, high usage of MSG in ethnic cuisines does not represent a situation in which intakes might achieve unsafe levels, even among individuals claiming idiosyncratic intolerance of such foods (Surrey, United Kingdom; 11). In one of the earlier studies indicating the presence of a reaction to MSG (12), patients first were exposed to a very large 5 gram dose of the isolated compound (as mentioned above, one could eliminate the erratic and minimal signs of reactions in careful testing when MSG is given with food). It was reported that the threshold dose (minimum amount) to get reactivity to MSG on rechallenge was 2.5 grams (still a huge amount for a single ingestion). The authors of the study concluded that the symptom characteristics did not support a usual allergy-based reaction (IgE-mediated mechanism). Thus, while some evidence of reaction was found in this study, it was only with huge doses given without food; the purported allergy-basis of the reaction, which might explain claimed reactions to the small amounts present in some meals, was not apparent. Proponents of the idea that there is widespread occurrence of an MSG-syndrome and overt MSG toxicity question the validity of all of the research indicating its safety, suggesting that information about adverse reactions is suppressed (18). Nonetheless, diverse international efforts repeatedly find little or no reaction, especially when MSG is combined with food (the method of ingestion that is the basis for the claimed adverse responses). A lack of adverse reaction to glutamate, at least for most people, makes biological sense when one considers the high amounts of glutamate already in the body and its ready availability from foods as they are digested. Since glutamate is present in large concentrations in the tissues of the small intestine, where MSG and other dietary glutamate is absorbed, it is not surprising that a reaction to glutamate is elusive. In hospitalized persons who require IV nutrition, glutamate is often included in the nutrition mix to assure maintenance of normal blood levels of this nutrient; adverse reactions are not reported from that method of administration either. Glutamate in Neurological DiseasesThe other concern about glutamate is related to its essential role as a neurotransmitter. The levels of glutamate in the central nervous system (brain and spinal cord) are highly regulated, since the neurons have sensitive receptors for the compound. In some neurological diseases, it is found that glutamate levels in the central nervous system become unusually high at sites of pathology. This can occur, for example, if the rate of degradation of glutamate is slowed by an impairment of the enzymes that are involved. Also, glutamate is excreted by immune cells that take part in inflammatory processes; the result is high local concentrations at the neurons in progressive neurological diseases such as MS and ALS. Glutamate levels in the central nervous system can also increase when the blood brain barrier is substantially weakened, as occurs after neurological surgery. The excess glutamate at the neuron acts as a poison; at high enough levels, the nerves exposed to glutamate can be completely and permanently damaged, so that they are no longer capable of transmitting signals. Thus, while glutamate is a major component of the body, and an essential part of the nervous system, high levels localized in the nerve cells can be quite toxic, and this is readily demonstrated in animal models. Laboratory research has revealed that in the progressive, debilitating disease ALS, one of the many processes involved in disease progression appears to be damage of nerve cells by accumulation of glutamate. In relation to multiple sclerosis, changes in control of glutamate homeostasis in the central nervous system might contribute to demyelination of the white matter of the brain (19). Based on preliminary animal studies, it has been suggested that glutamate dumped by immune cells can exacerbate the nerve damage (20). One of the means by which a stroke (causing blockage of blood circulation to the brain) results in brain damage is through an increase in glutamate levels in the brain cells (of course, oxygen deprivation and other effects are also contributors). These findings point to local glutamate excess as an important factor in brain diseases. The role of glutamate in neurological disorders has raised the question as to whether persons with such neurological diseases might have to be careful not to get high levels of either glutamine or glutamate via their diet and/or by taking glutamine supplements. Since glutamine is the main amino acid in the body and is produced by the body in response to deficiency, it is probably not possible to significantly lower the glutamine levels from the levels that already exist at the time of diagnosis. Indeed any attempt to significantly lower glutamine levels could lead to numerous adverse consequences. However, one can avoid excessive intake of glutamate by minimizing ingestion of foods containing MSG and hydrolyzed vegetable protein, and by limiting the dosage of any glutamine supplementation. Glutamate levels in the blood increase slightly with high doses of supplemental glutamine administration (e.g., 15 grams in a single dose), but not with moderate doses (e.g., 5 grams in a single dose). It is not known whether modification of dietary intake of glutamine and glutamate will have any effect on the degenerative brain disorders, since the mechanisms by which high glutamate content occurs in the brain may not be dependent on blood glutamate levels. However, the situation remains unclear and questions have been raised for the case that would most likely involve alterations in blood levels: weakness of the blood brain barrier after surgery, which would allow the blood glutamate to influence the central nervous system levels and potentially exacerbate the brain edema that occurs (21). Until more is known about glutamine supplementation in relation to these brain diseases, it is recommended that patients who have nerve-damaging, chronic neurological diseases, such as ALS and multiple sclerosis, and those who have had recent neurological surgeries (such as for brain tumors) limit their intake of supplemental glutamine. A modest glutamine supplement level of about 5-10 grams/day is likely to have some benefit in relation to muscle wasting, immune responsiveness, or intestinal disorders, without promoting increased glutamate levels in the blood. Even 10 grams of glutamine is a small amount compared to the total body reserves that are already present, and studies show no significant glutamate increases in the blood after consuming such amounts. The higher levels of glutamine administration, commonly in the range of 15-30 grams per day, might be reserved for patients who do not suffer from these neurological diseases. Patients with neurological diseases who wish to avoid high glutamate levels should stay away from the meals that contain added glutamate in any large amount; but, as with glutamine, any attempt at total glutamate avoidance (including the form of glutamic acid in all protein foods) is neither possible nor necessary. The glutamate levels of the blood will be maintained at a certain level by normal metabolism even without adding dietary glutamate, but can be increased significantly only by consuming large amounts of the amino acid. There may be specific chemical inhibitors of glutamate synthesis or glutamate uptake in neurons that would have a dramatic impact on the neurological diseases. It has been proposed that some drugs used experimentally to prevent brain damage from strokes (that function by inhibiting glutamate in neurons) might be of value in this regard. SUMMARYGlutamine is an important body component that helps deal with several stressful situations, including the consequences of chemotherapy, burns (including radiation therapy burns), and injuries (including surgical injuries). Glutamine levels will naturally be high in persons who maintain a large muscle mass and ingest plenty of protein in their diet, but can become critically low in those who have little muscle mass (especially with muscle-wasting disease conditions) and limited dietary intake of protein. Glutamine supplementation appears to be an inexpensive, convenient, and potentially helpful means of promoting healing in those who are undergoing cancer therapies, suffering from intestinal diseases and surgeries, and recovering from injuries. Similarly, glutamate is an important body component; it acts as a neurotransmitter, is part of the key antioxidant system glutathione, and is important for the transfer of ammonia. As with glutamine, it is present in adequate quantities in those with good muscle mass and a reasonably high protein diet. Glutamate is incidentally added to some foods in the form of MSG and hydrolyzed protein flavorings, and there have been concerns raised that this leads to adverse allergy-type reactions. Thus far, a great body of research has shown that such reactions either do not exist, or are very rare and not allergy-type reactions. Therefore, fear of this food component is usually unjustified. Glutamate at high enough levels in the central nervous system can cause nerve damage; these high levels are mainly the result of unusual situations such as autoimmune attacks against nervous system components (as occurs with MS and ALS) or following brain surgery. Since glutamine is converted to glutamate, supplementing glutamine at very high levels in persons who have such neurological disorders may be contraindicated. Therefore, glutamine supplementation in such individuals, if called for, should be limited to about 5-10 grams per day until more is known; high glutamate meals should also be avoided in this situation. REFERENCES
|